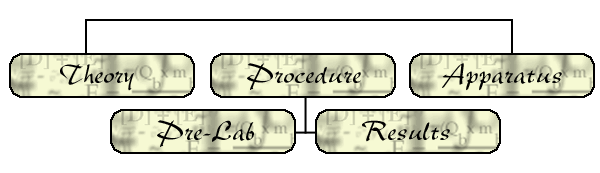
Calorimeter
cross-section
cross-section
Bomb
cross-section
cross-section
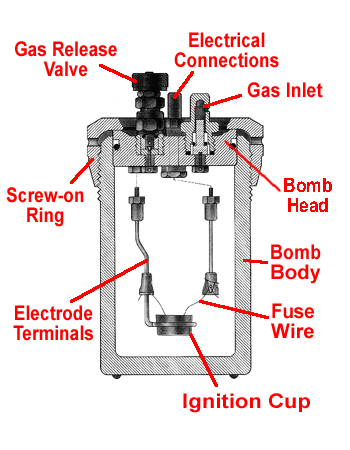
The metal bomb provides a constant-volume system in which the combustion will reaction take place. The sample pellet is placed in the ignitiona cup and the fuse wire is carefully arranged to touch the pellet but not the cup. The bomb is sealed by screwing the cap on and then filled with a high pressure of pure oxygen. The electrical connections are made at the top and the bomb is placed into the water bucket.